|
The reaction of metals with salt salutations:ĭisplacement reaction is the reaction of metals with other metallic salt solutions. Magnesium hydrochloric acid magnesium chloride hydrogenĤ. Magnesium chloride and hydrogen are formed when magnesium reacts with dilute hydrochloric acid. Zinc hydrochloric acid zinc chloride hydrogenī. Zinc reacts with dilute hydrochloric acid to give zinc chloride and hydrogen gas. 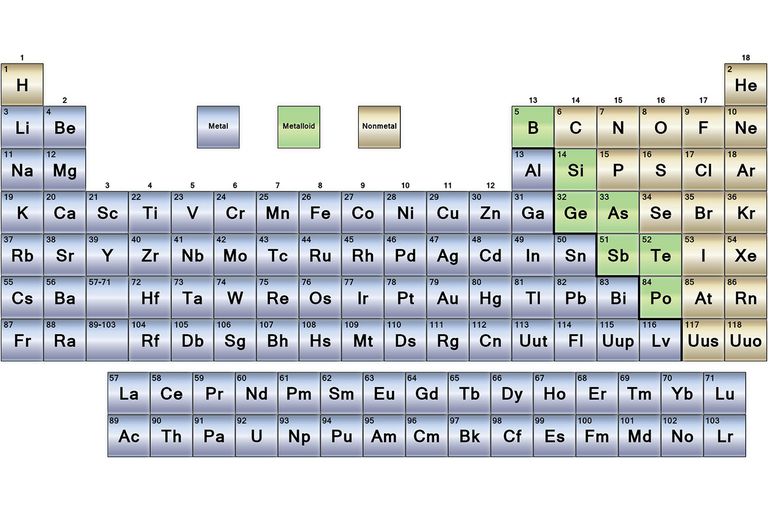
When metals react with dilute acid then metal salt and hydrogen gas are formed.Ī. Copper, silver, gold do not react with steam.ģ. Other metals do not react with water or very slowly reacts. Magnesium reacts with water and forms magnesium hydroxide and hydrogen gas. Calcium forms calcium hydroxide and hydrogen gas when it reacts with water.Ĭ. Sodium forms sodium hydroxide and hydrogen gas along with a lot of heat when reacting with water.ī. Metals form hydroxide and hydrogen gas when it reacts with water.Ī. Silver, gold, and platinum do not combine with the oxygen of air even at high temperatures. Potassium forms potassium oxide when it reacts with oxygen. Sodium forms sodium oxide when reacts with oxygen. The metal oxide is formed on the reaction of metals with oxygen.Ī. Mercury is liquid at room temperature (1) & (3). Most of the metals like iron, copper, aluminum, silver, etc. Metals are solid at room temperature, except mercury. The shiny appearance of metals makes them useful in making jewelry. The property of metal of having a shining surface is called “metallic luster”. Metals have the quality of reflecting light from its surface. Tungsten has the highest melting point and silver has a low boiling point. Metals have high melting points and boiling points. So the reason electric wires are made of metals like copper and aluminum. Metals have good conduction of heat and electricity. Iridium and osmium have the highest density and lithium have the lowest density. Metals have high density and are very heavy. Copper and aluminum metals are very ductile. Some metals are more ductile than others. 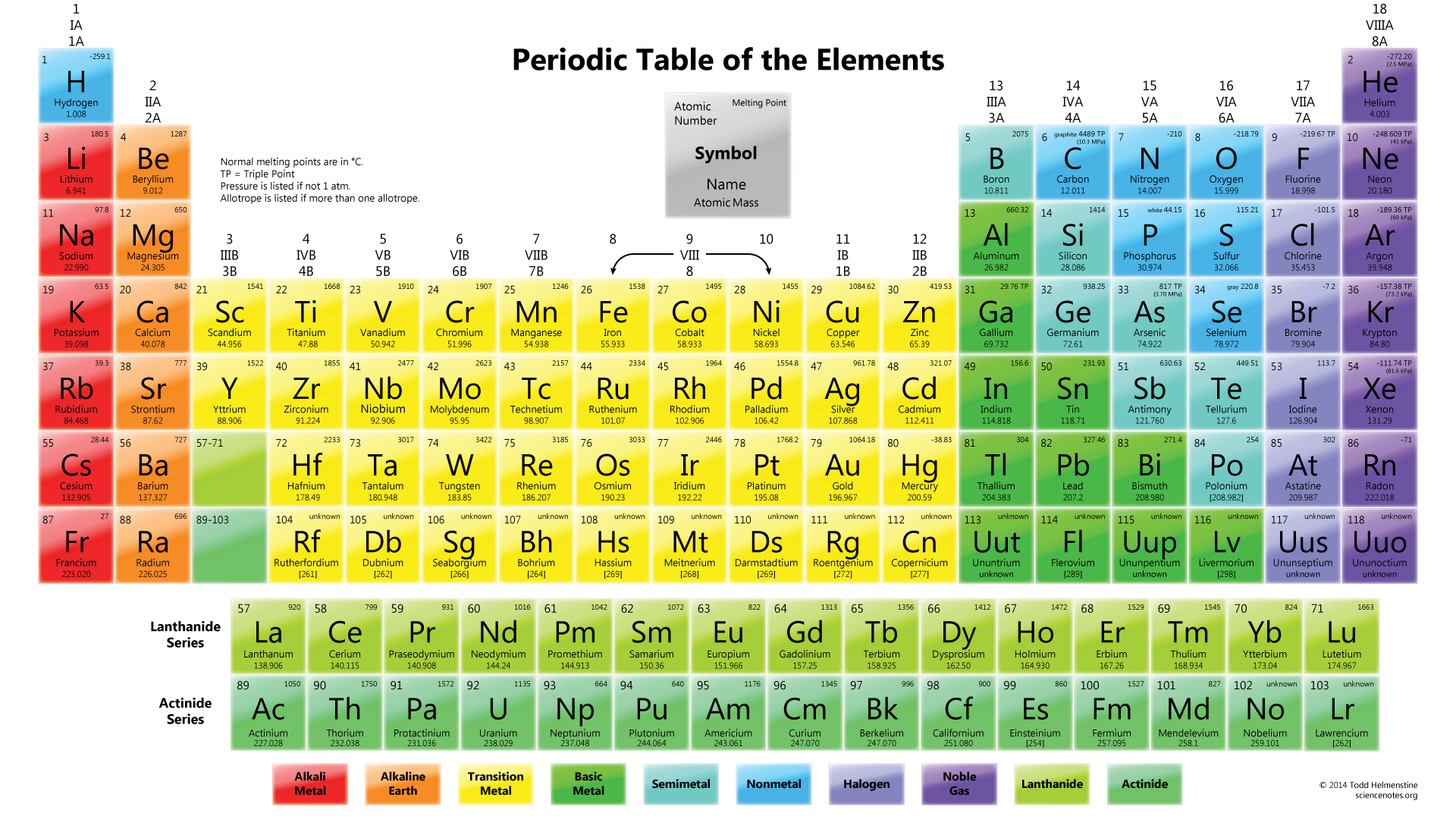
But all the metals are not equally ductile. It is another important property of metals. Most of the metals are strong and have high tensile strength. Because of the property, iron is used in making big ships. That is, metals can be beaten into thin sheets with a hammer. Malleability is an important characteristic of metals. These metals are very soft and can be cut with a knife. Most of the metals in the periodic table are hard, except sodium, potassium, lithium, etc. All these metals are solid, except mercury which is liquid (3). 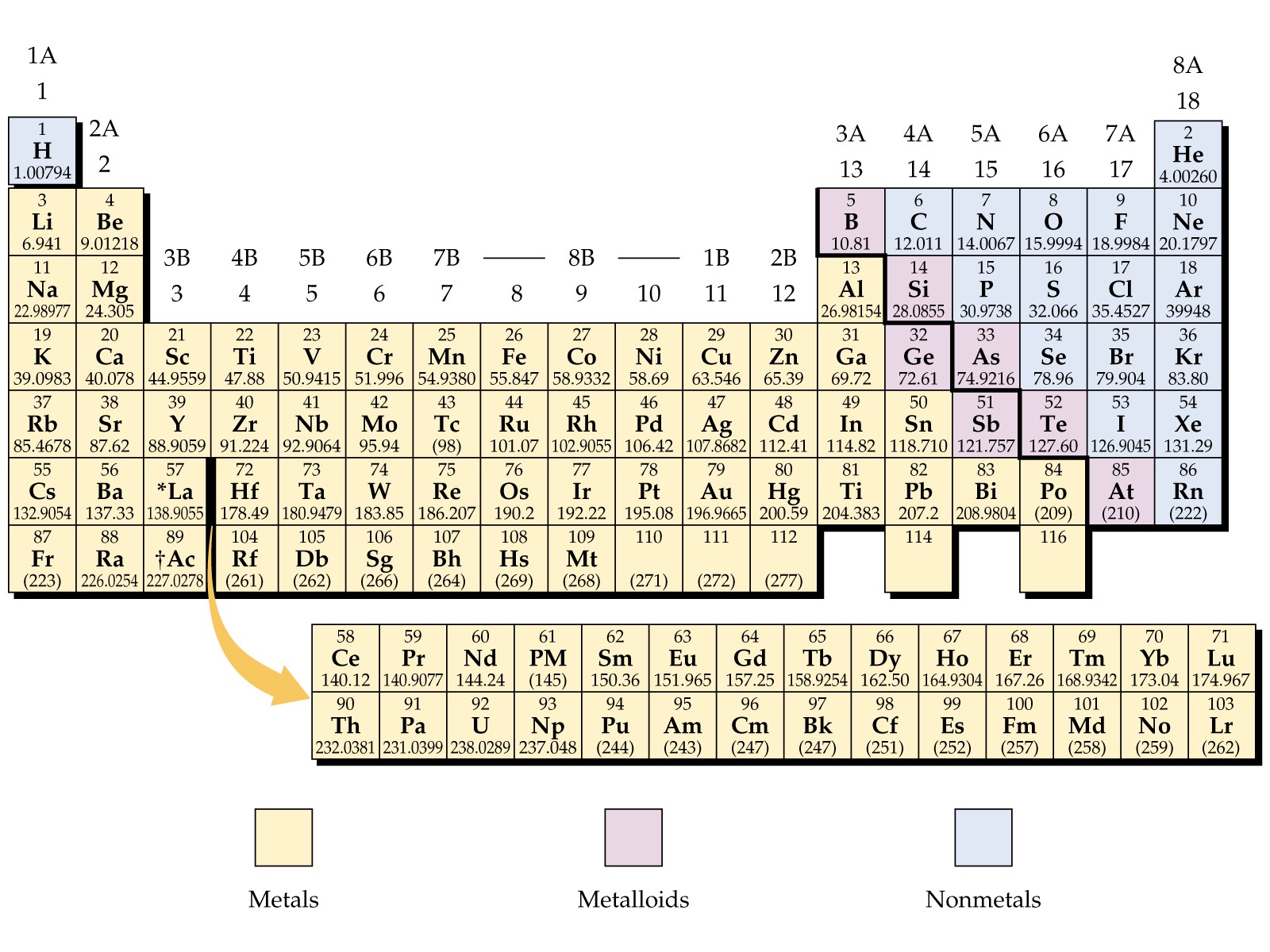
Some of the examples of metals are silver, copper, gold, platinum, zinc, iron, aluminum, mercury, lead, magnesium, etc. When reacts it usually produces alkaline oxides (3). Also relatively heavy at a weight and produces metallic sound when hit. These are capable of bright, shiny, and light reflection. Metals are those elements that are hard, strong in nature. Metals definition, examples and properties Definition of metals The metal and non-metal on the periodic table are discussed below (1) & (3). From the sophisticated electronic devices, the study of metals has gained a lot of importance in daily life and their wide use. The elements on the right side of the periodic table are non-metals. More than 75% at the left and middle part of the periodic table are metal. Besides these, there is another section of elements called as a metalloid. All of these elements are broadly classified as metal and non-metal on the periodic table. The periodic table contains a total of 118 elements so far. Metal and non-metal on the periodic table.Difference between metal, non-metal, and metalloids (1) & (3).Metalloids definition, examples and properties.Non- metal definition, examples and properties.Metals definition, examples and properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
